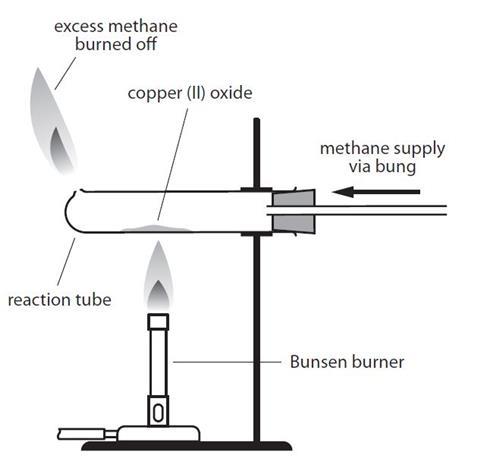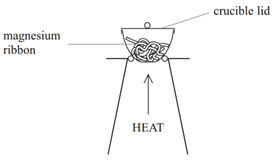
1:36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide) - TutorMyself Chemistry

SOLVED: Q2 Calculate the average atomic mass of magnesium using the following data for three magnesium isotopes. You must show all your work to get credit Isetope IaSS (W)) relativg abundance Mg-24

Question Video: Calculating the Mass of Calcium Carbonate Required to Produce a Given Mass of Calcium Oxide | Nagwa

Zinc oxide reacts whith carbon, on heating, to from zinc metal and carbon monoxide. Write a balanced - YouTube

Reactions of Group 2 Oxides, Hydroxides & Carbonates (2.2.2) | CIE A Level Chemistry Revision Notes 2022 | Save My Exams

Carbon dioxide is added to 112 g of calcium oxide. The product formed is 200 g of calcium carbonate. Calculate the mass of carbon dioxide used. Which law of chemical combination will

Metal carbonates are known to undergo thermal decomposition, producing the metal oxide and releasing carbon dioxide. The process is described by the following generic equation, in which M represents an unknown divalent