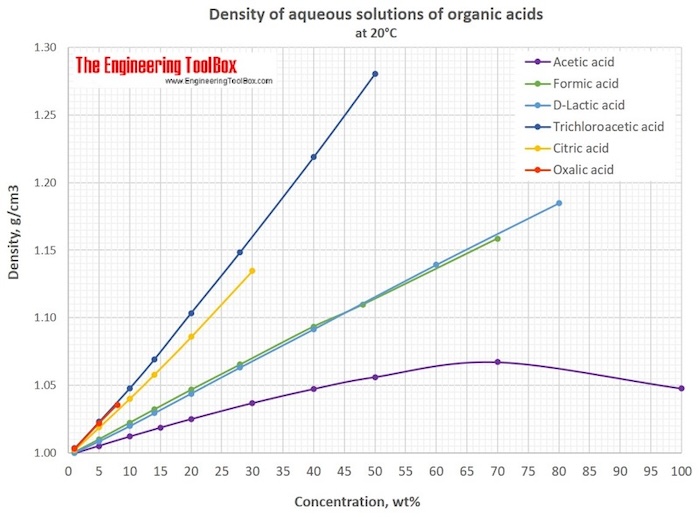
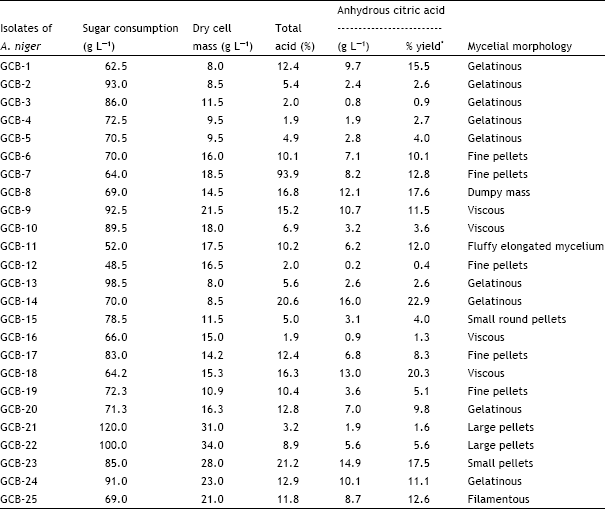
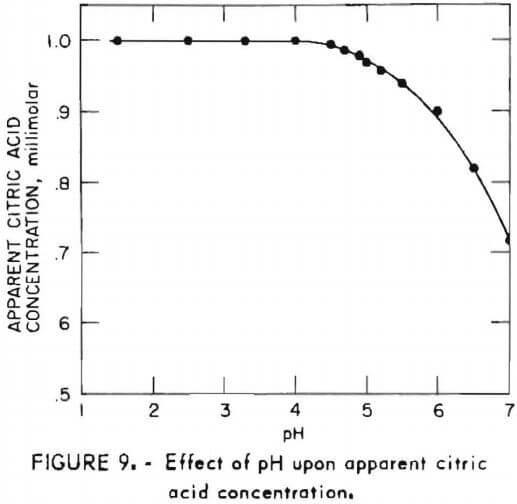
DETERMINING THE CONCENTRATION OF CITRIC ACID IN A SOFT DRINK USING ACID/BASE TITRATION INTRODUCTION The acid content of many foo

The effect of the concentration of citric acid and pH values on the preparation of MgAl2O4 ultrafine powder by citrate sol–gel process - ScienceDirect

Effect of citric acid on the acidification of artificial pepsin solution for metacercariae isolation from fish - ScienceDirect

Lemon juice normally has a pH of 2. If all the acid the lemon juice is citric acid and there are no citrate salts present, then what will be the citric acid
FTIR spectroscopy as a unified method for simultaneous analysis of intra- and extracellular metabolites in high-throughput screening of microbial bioprocesses | Microbial Cell Factories | Full Text

Lemon juice contains citric acid with a concentration of approximately 0.30 M (around 6% by mass), giving rise to its acid properties. Citric acid is generally considered a triprotic acid (capable of
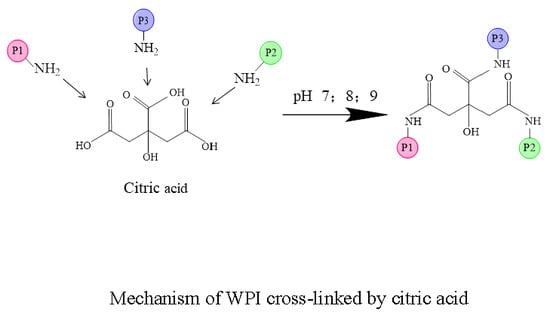
Molecules | Free Full-Text | Surface Hydrophobicity and Functional Properties of Citric Acid Cross-Linked Whey Protein Isolate: The Impact of pH and Concentration of Citric Acid

SOLVED:Citric acid, which is present in citrus fruits, is a triprotic acid (Table 16.3) . (a) Calculate the pH of a 0.040M solution of citric acid. (b) Did you have to make

Determination of Citric acid in Soft drinks, Juice drinks and Energy drinks using Titration | Semantic Scholar