Aqueous aggregation behavior of citric acid coated magnetite nanoparticles: Effects of pH, cations, anions, and humic acid | Semantic Scholar

Production of Citric Acid by <i>Candida lipolytica</i> under Fermentation Conditions Using a Plackett-Burman Design

Destructive effects of citric acid, lactic acid and acetic acid on primary enamel microhardness | Semantic Scholar
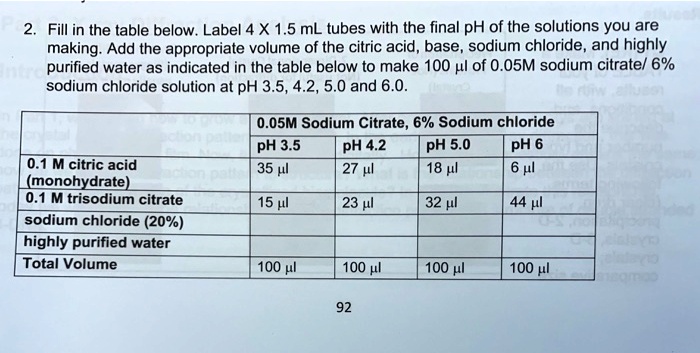
SOLVED: Fill in the table below Label 4 X 1.5 mL tubes with the final pH of the solutions you are making: Add the appropriate volume of the citric acid, base, sodium

Improving Clarity and Stability of Skim Milk Powder Dispersions by Dissociation of Casein Micelles at pH 11.0 and Acidification with Citric Acid | Journal of Agricultural and Food Chemistry

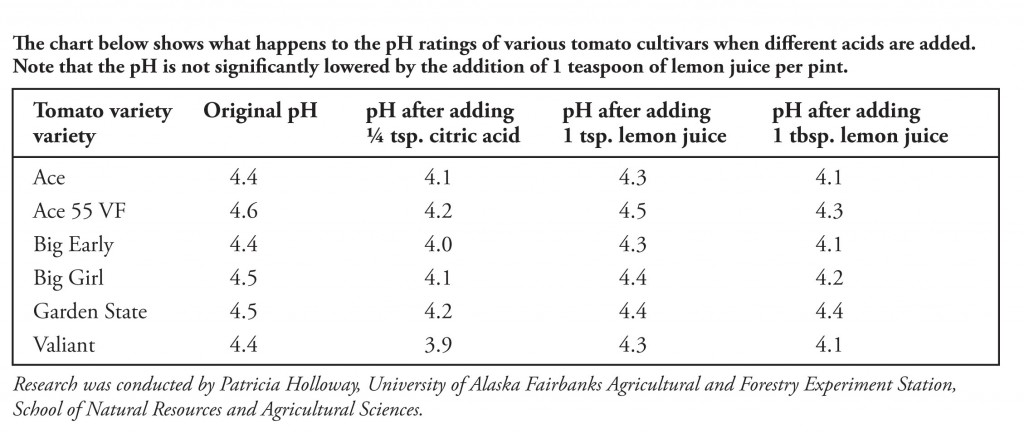
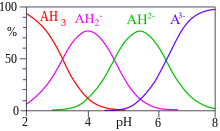
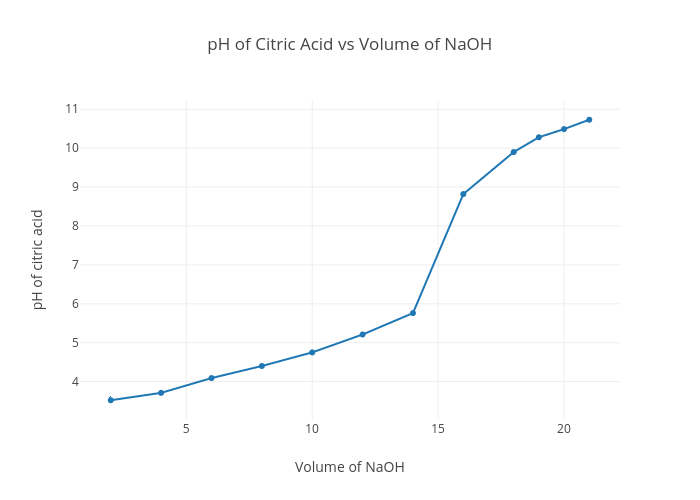
Citric acid is a triprotic acid with Ka values of 3.14, 5.95, and 6.39. Estimate the pH of a 0.15 M solution of a monosodium salt. | Homework.Study.com