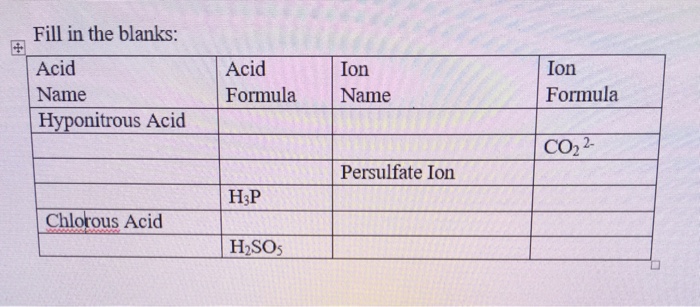
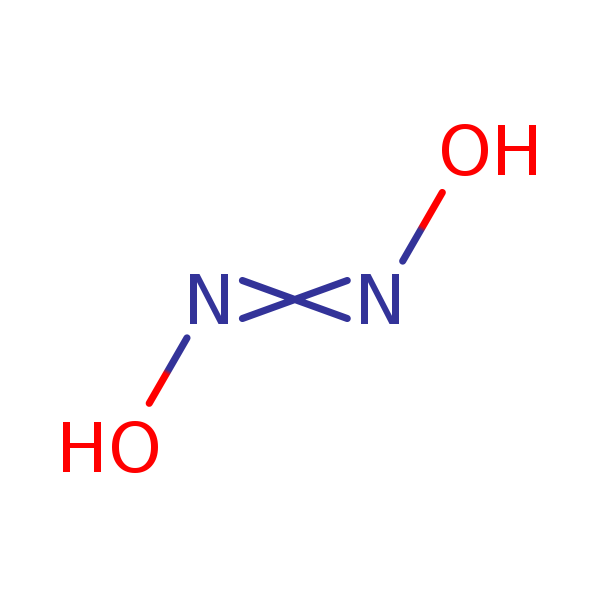
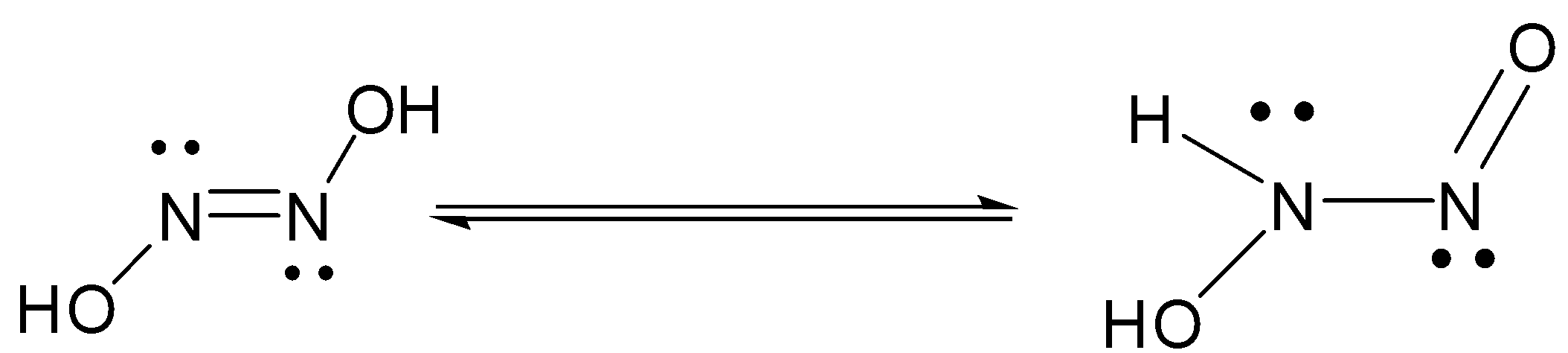
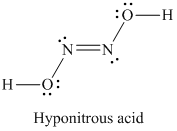
Two substances with empirical formula HNO are hyponitrous acid (M = 62.04 g/mol) and nitroxyl (M = - Brainly.com
Solved: Chapter 21 Problem 101AP Solution | Onekey Blackboard, General Chemistry 4th Edition | Chegg.com
Give the formula and the oxidation state of nitrogen in the following acids. (i) Hyponitrous acid, (ii) Nitrous acid, - Sarthaks eConnect | Largest Online Education Community

799. The mechanism of the oxidation of nitrous acid by hyponitrous acid. Part I - Journal of the Chemical Society (Resumed) (RSC Publishing)




![Hyponitrous acid] Hyponitrous acid]](https://www.degruyter.com/document/doi/00.0000/IUPAC.iupac.compound.61744/asset/images/61744.png)