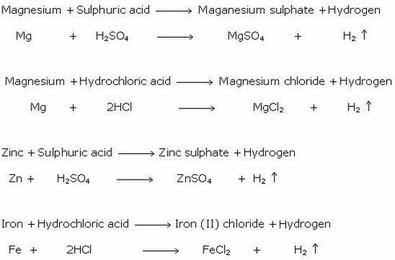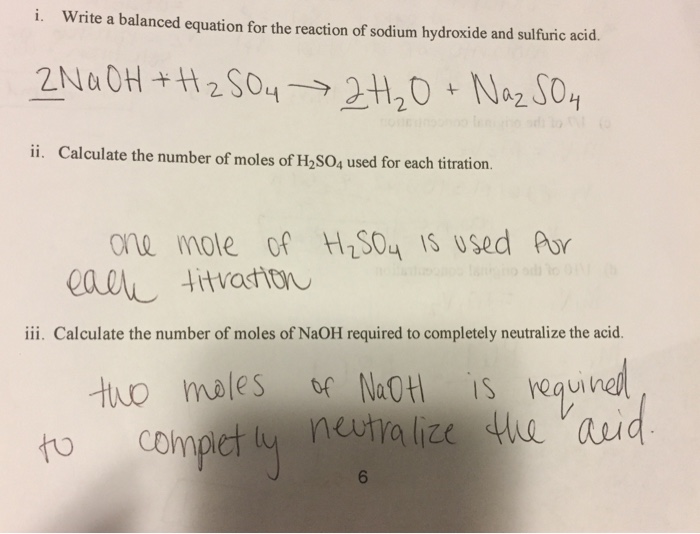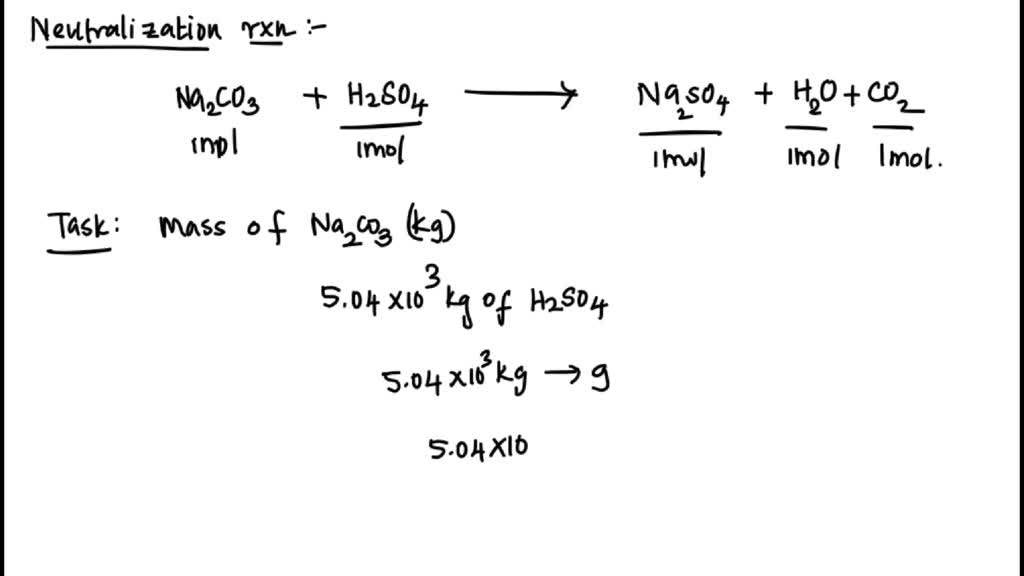
SOLVED: Sodium carbonate (Na2CO3Na2CO3) is used to neutralize the sulfuric acid spill. How many kilograms of sodium carbonate must be added to neutralize 5.04×103 kgkg of sulfuric acid solution?

Neutralization Reaction: Determine Molarity of a sulfuric Acid Solution when Neutralized by NaOH - YouTube

Neutralization curve of Mg(OH)2 (MH) with a sulfuric acid solution (0.4 M). | Download Scientific Diagram
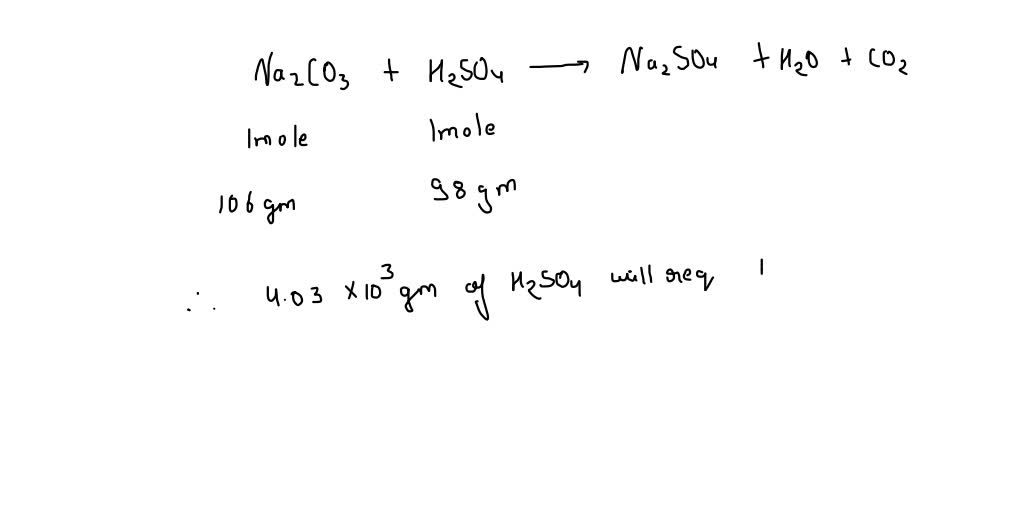
SOLVED: Sodium carbonate (Na2CO3) is used to neutralize the sulfuric acid spill. How many kilograms of sodium carbonate must be added to neutralize 4.03×103 kg of sulfuric acid solution? Express your answer

Question Video: Calculating the Volume of Sulfuric Acid That Completely Neutralizes a Given Volume and Concentration of Sodium Hydroxide | Nagwa

KR100847444B1 - Method for neutralizing sulphuric acid waste water by using calcined dolomite - Google Patents